Two oppositely charged particles and their electric fields (with arrows pointing in the direction of the force) the proton is left and the electron is right. Thus, the forces between particles are a result of the electric fields created by all the particles individually. These fields can be thought of as a particle warping space around it, and this field then interacts with other particles. Protons and electrons have an another intrinsic property called charge. A proton’s electric field is directed outward, while an electron’s field is directed inward. 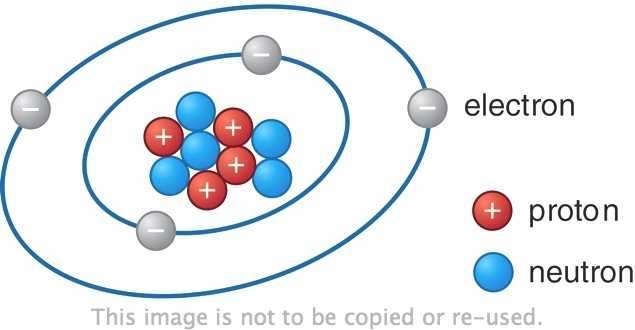
Particles create an electric field, even with no other particles around.This means that the further apart two particles are, the less force they exert on each other. The force, like gravity, is inversely proportional to the square of the distance. Protons and neutrons constitute the bulk of the mass of atoms. The mass of a proton or a neutron is about 1836 times greater than the mass of an electron. Neutrons have approximately the same mass as protons but no charge. The same number of electrons is required to make 1.00 C of electric charge. Relative charges of 1 and +1 are assigned to the electron and proton, respectively. The number n of protons required to make +1.00 C is. The magnitude of the electric forces that particles exert on each other depends on the distance. and the electron carries 1.602 × 10 19 C. 
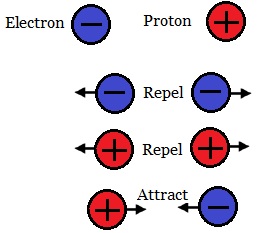
Opposite charges (like a proton and electron) attract, while like charges repel.Interestingly, even holes are treated as charge carriers in solar physics, and they possess a positive charge. Later we will discuss the hole, which is a vacancy left in an orbital when an electron jumps to a higher energy state. Both protons and neutrons have a mass of 1, while electrons have almost no mass. Charge is quantized as a multiple of the electron or proton charge: proton charge: e1.602 x. The negative charge of one electron balances the positive charge of one proton. one proton or electron is called the elementary charge e. The charge of a proton is e, or the same number but positive, while a neutron has no charge. As summarized in Table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. Electrons have a charge commonly denoted as –e, or -1.602 x 10 -19 Coulombs (C). Fundamental particles such as electrons have associated with them a specific electric charge, or intrinsic property that determines the Coulomb forces or electromagnetic force they exhibit on each other. To discuss electricity, we must introduce the concept of charged particles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
